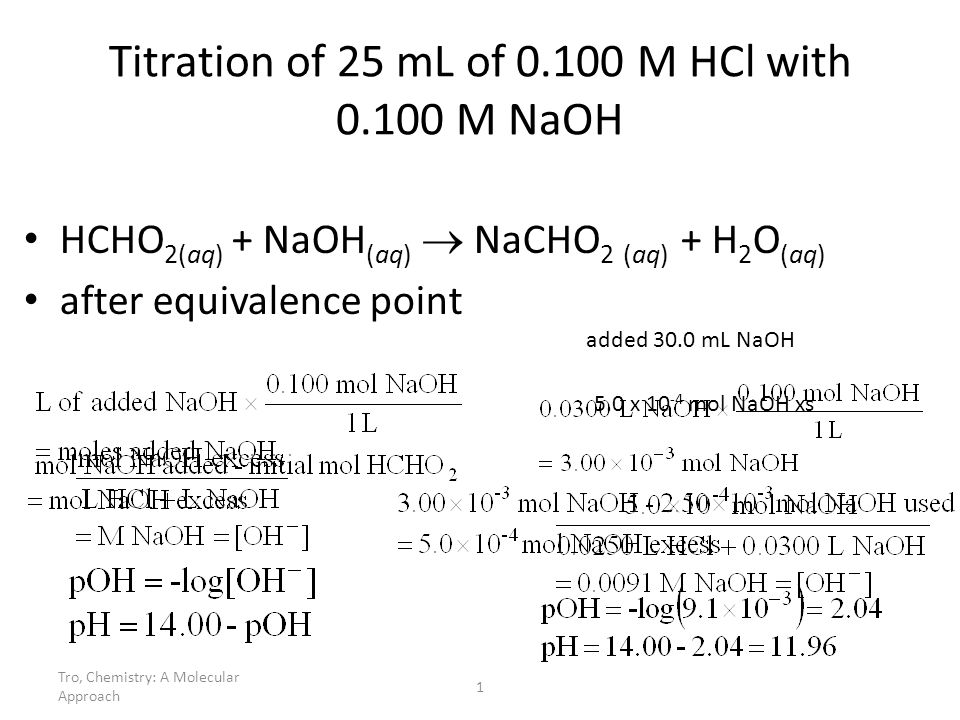
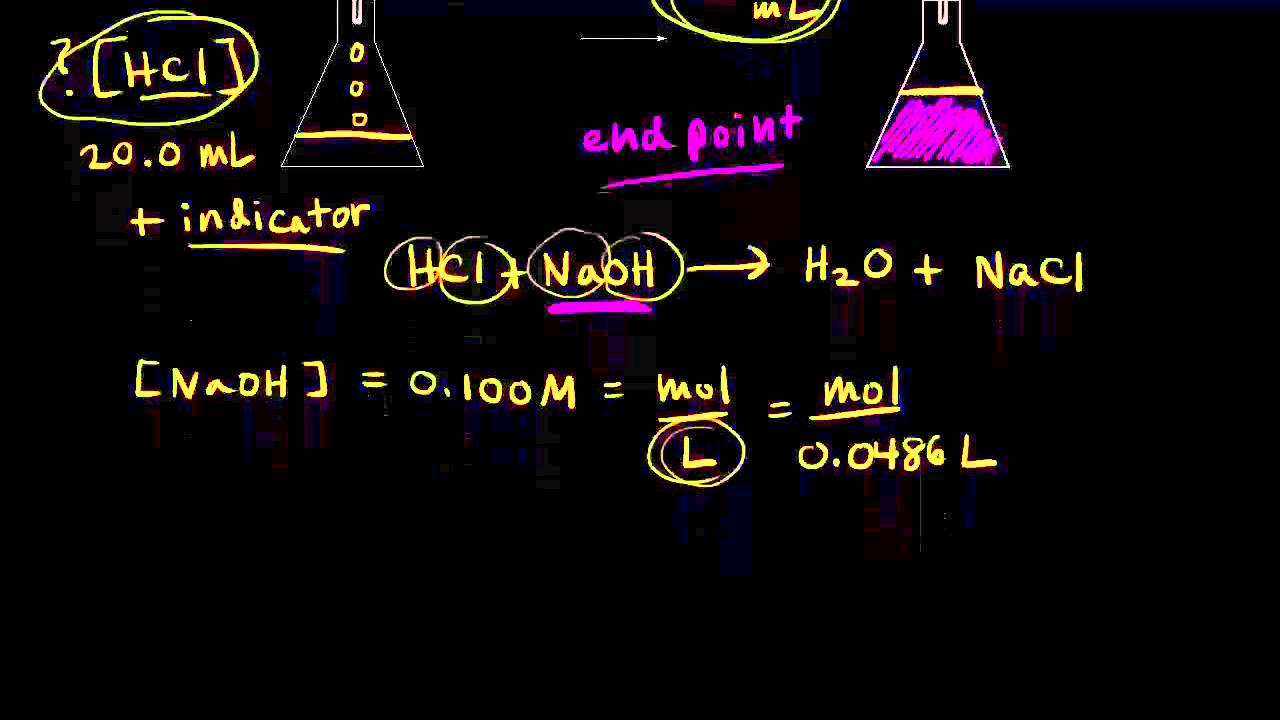
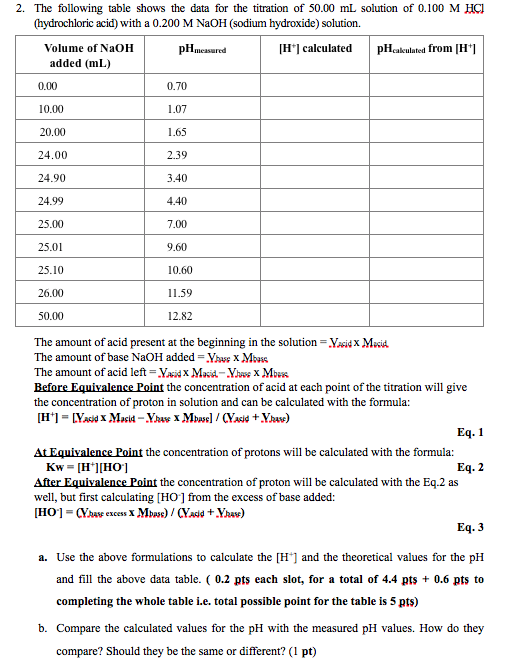
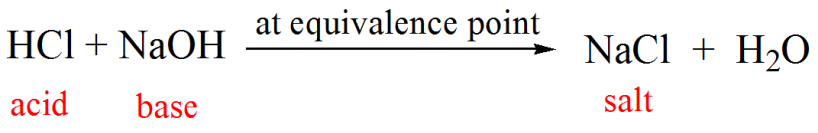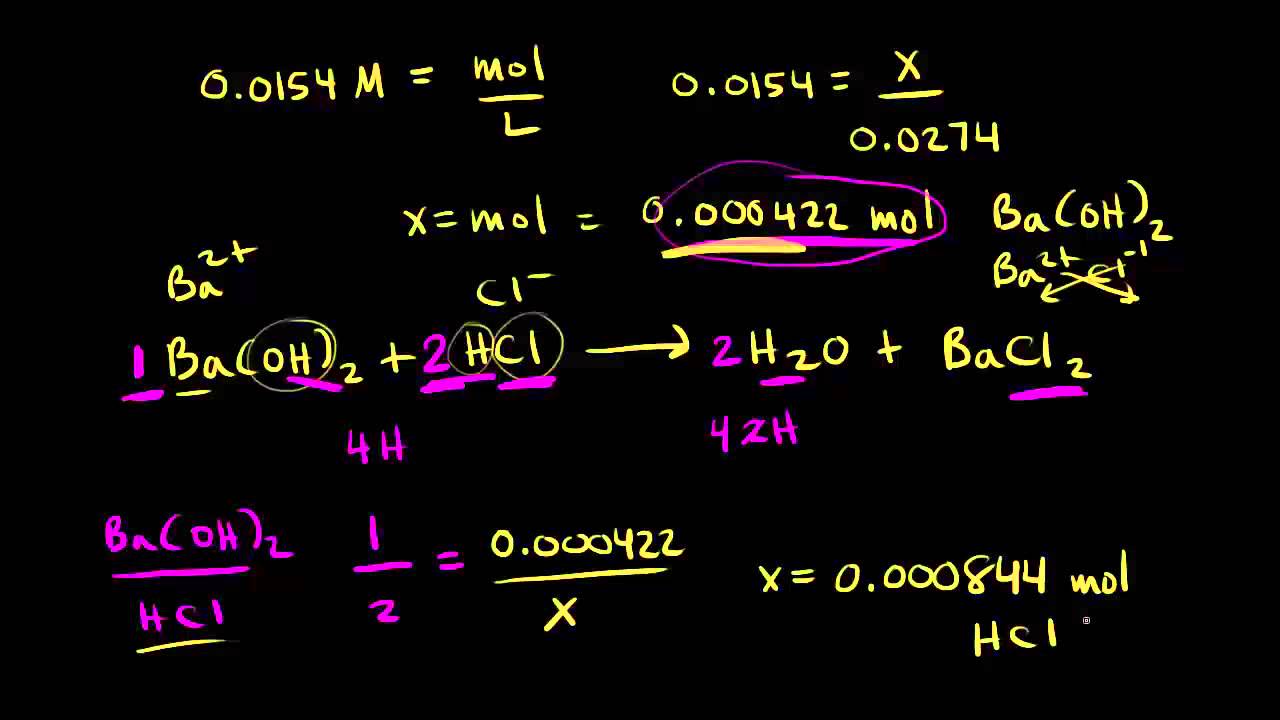Titration of Sodium Hydroxide with Hydrochloric acid | FSC107 - General Chemistry Laboratory - XMUM | Thinkswap

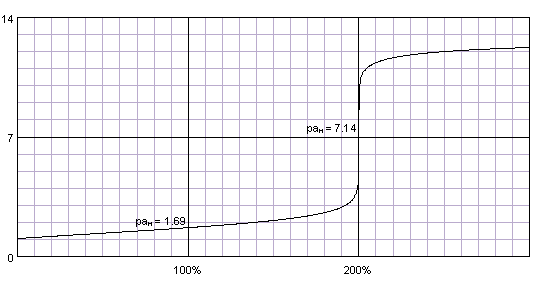
Lab VIII – Titration of Weak (CH3COOH) and Strong (HCL) Acids via Strong Base (NaOH) | nmiller17chem
A 10.0 mL sample of 0.250M HCl (aq) is titrated to the end point with a 14.0 ml sample of NaOH(aq). What is the molarity of the NaOH (aq)? - Quora

Calculate the concentration of HCl acid if 50 ml of HCl is required to neutralize 25 ml of 1 M NaOH in acid base titration.

SOLVED: Molarities of Solution:: Solution Molarity NAOH LlolsM HCI MldleH HC,HO: L10XM Titration of Hydrochloric Acid: mL base needed to reach the equivalence point: 44Im Show how you detenined this volume from